United States In Vivo CRO Market Trends and Future Forecast 2034
The in vivo contract research organization sector is witnessing significant expansion as pharmaceutical and biotechnology companies continue to increase investments in drug discovery, preclinical testing, and advanced therapeutic development. Rising demand for outsourced research services, combined with growing pressure to accelerate clinical pipelines, has strengthened the role of specialized research organizations across global healthcare ecosystems. Increasing focus on oncology, autoimmune disorders, and gene therapies is also contributing to the rapid evolution of this industry.
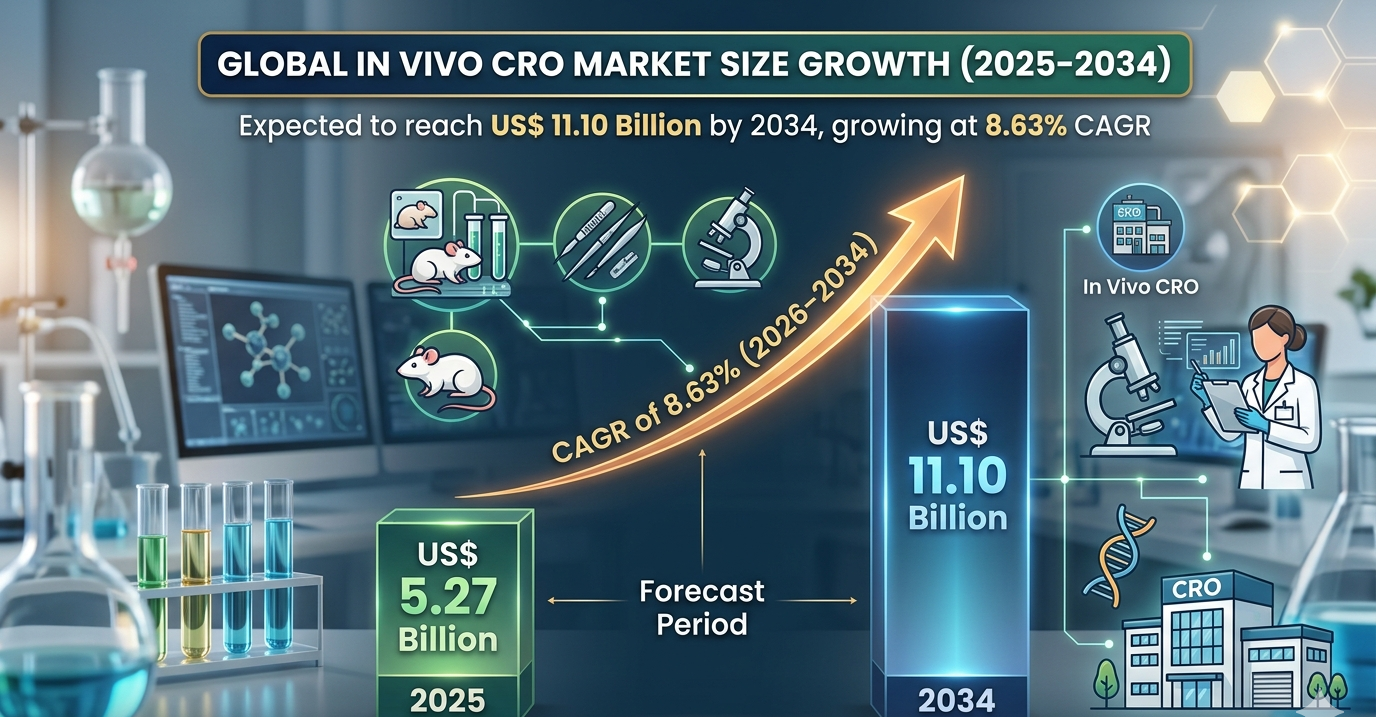
The In Vivo CRO Industry Size is projected to grow substantially over the forecast period, supported by increasing research complexity and expanding biopharmaceutical innovation. According to industry estimates, the sector was valued at US$ 5.27 billion in 2025 and is expected to reach US$ 11.10 billion by 2034, registering a CAGR of 8.63% during 2026–2034. The growing adoption of outsourced preclinical testing services and advancements in personalized medicine are expected to remain central growth drivers.
Get Sample PDF @ https://www.theinsightpartners.com/sample/TIPRE00007238
Rising Demand for Drug Discovery and Development
The increasing global burden of chronic and rare diseases has accelerated the development of novel therapeutics, thereby driving demand for in vivo CRO services. Pharmaceutical companies are under pressure to shorten development timelines while ensuring regulatory compliance and maintaining research quality standards. As a result, outsourcing preclinical and toxicology studies to specialized CROs has become a preferred operational model.
In vivo CROs provide essential services such as pharmacokinetic studies, toxicity testing, efficacy analysis, and animal model research. These services are crucial during early-stage drug development and help pharmaceutical companies reduce infrastructure investments and operational costs. The growing pipeline of biologics, biosimilars, and targeted therapies further strengthens demand for specialized research support.
The increasing number of therapies under development for oncology, cardiovascular diseases, CNS disorders, and autoimmune conditions is expected to sustain long-term industry expansion. In addition, pharmaceutical sponsors are increasingly relying on CROs to enhance productivity and improve research efficiency.
Technological Advancements Transforming Preclinical Research
Rapid technological advancements are reshaping the operational framework of the in vivo CRO industry. Artificial intelligence, machine learning, and advanced analytics are increasingly being integrated into preclinical workflows to improve study design, predictive modeling, and data interpretation.
AI-enabled tools help researchers analyze complex biological datasets more efficiently while improving reproducibility and reducing experimental errors. These technologies are also supporting faster identification of potential drug candidates and optimizing research outcomes.
Another major trend influencing industry development is the adoption of 3D cell culture systems and organ-on-a-chip technologies. These advanced testing models provide more accurate simulation of human physiological responses compared to conventional animal models. Such innovations improve drug toxicity assessment and therapeutic efficacy analysis while supporting ethical research practices.
The growing use of non-rodent models, including rabbits, pigs, and non-human primates, is also contributing to higher-quality preclinical data generation. These models are increasingly preferred for oncology and immunology studies due to their closer physiological similarity to humans.
Expansion of Personalized Medicine and Gene Therapy
The rapid growth of personalized medicine and gene therapy research is creating substantial opportunities for in vivo CRO providers. Advanced biologics and cell-based therapies require sophisticated in vivo testing to evaluate long-term safety, efficacy, and immune responses before entering clinical trials.
Gene therapy development, in particular, has emerged as a major area of investment across the healthcare sector. Therapies targeting rare genetic disorders, neurological conditions, and inherited diseases require highly specialized testing capabilities. CROs with expertise in gene editing technologies and customized in vivo models are increasingly gaining strategic importance.
Patient-derived xenograft models and disease-specific animal models are becoming essential tools in precision medicine research. These tailored models allow pharmaceutical developers to better predict patient responses and improve clinical trial success rates.
The expansion of biotechnology companies across North America, Europe, and Asia Pacific is further supporting demand for outsourced in vivo research services. Increasing collaboration between pharmaceutical manufacturers and CRO providers is expected to accelerate innovation throughout the forecast period.
Regulatory Environment and Industry Challenges
Regulatory agencies worldwide continue to enforce stringent safety and efficacy standards for drug approvals. Organizations such as the FDA and EMA require extensive preclinical testing before therapies can advance to human clinical trials. This regulatory pressure is increasing reliance on CROs that possess expertise in GLP-compliant testing and documentation.
At the same time, ethical concerns regarding animal testing remain a significant industry challenge. Regulatory authorities and research institutions are encouraging the development of alternative testing approaches that reduce animal usage while maintaining scientific accuracy. This trend is accelerating investments in organ-on-a-chip technologies and advanced in vitro systems.
Data management complexity, rising operational costs, and evolving compliance requirements also present challenges for industry participants. However, CRO providers that invest in digital transformation, automation, and specialized therapeutic expertise are expected to maintain competitive advantages.
Regional Insights and Competitive Landscape
North America continues to dominate the global in vivo CRO industry due to strong pharmaceutical research infrastructure, high R&D spending, and the presence of leading biotechnology firms. The United States remains a major contributor to industry revenue because of increasing investments in drug development and precision medicine.
Europe represents another significant regional segment supported by favorable regulatory frameworks and growing biopharmaceutical innovation. Countries such as Germany, France, and the United Kingdom are witnessing increased outsourcing of preclinical studies.
Asia Pacific is expected to record the fastest growth during the forecast period. Expanding pharmaceutical manufacturing capabilities, lower operational costs, and increasing clinical research activities in China and India are attracting global pharmaceutical companies to the region.
Key industry participants include:
- Charles River
- Covance Inc.
- WuXi AppTec
- Syneos Health
- GVK Biosciences Private Limited
- Iris Pharma
- MELIOR DISCOVERY
- Washington Biotechnology, Inc.
These organizations are focusing on acquisitions, technology integration, and service portfolio expansion to strengthen their global positions.
Future Outlook
The future of the in vivo CRO industry remains highly promising as pharmaceutical and biotechnology companies continue to prioritize research outsourcing and advanced therapeutic development. Growing investments in gene therapies, biologics, and precision medicine are expected to generate sustained demand for specialized preclinical testing services. Technological advancements including AI-driven analytics, organ-on-a-chip systems, and customized disease models will further improve research efficiency and predictive accuracy.
Related Report:
1) Compounding Pharmacies Market Growth and Analysis by 2028
2) Colorectal Cancer Diagnostics Market Key Players and Opportunities by 2028
3) Anti-Acne Cosmetics Market Overview, Growth, Trends, Analysis, Research Report (2021-2031)
About Us -
The Insight Partners provides comprehensive syndicated and tailored market research services in the healthcare, technology, and industrial domains. Renowned for delivering strategic intelligence and practical insights, the firm empowers businesses to remain competitive in ever-evolving global markets.
Contact Us:
Ankit Mathur | The Insight Partners
E-mail: [email protected]
Phone: +1-646-491-9876
Also Available in : Korean German Japanese French Chinese Italian Spanish
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Games
- Gardening
- Health
- Home
- Literature
- Music
- Networking
- Other
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness



